Table of Contents
Key Takeaways
- Glutathione Precursor 🔧: NAC provides cysteine, the rate-limiting amino acid for glutathione synthesis; reliably increases blood and brain GSH at adequate doses (≥1200-3000 mg/day)
- Spike Protein Relevance: NAC inhibits ferroptosis (iron-dependent cell death) triggered by spike protein in microglia; may protect against basal ganglia neurodegeneration; disrupts disulfide bonds in spike aggregates
- Neuroprotection Evidence: Early Parkinson's trials show ~13% UPDRS motor improvement + 4-9% dopamine transporter increase; Alzheimer's data weak/inconsistent for standalone use
- Protein Aggregate Claims: NO human evidence NAC "breaks up" amyloid, alpha-synuclein, or prion-like clumps; preclinical data only, benefits likely indirect via glutathione restoration
- BBB Protection: NAC's thiol groups may protect tight junction proteins from MMP-9 degradation and reduce neuroinflammation through GSH-mediated antioxidant effects
- Long COVID Reality: Limited human RCTs; not listed in major 2026 meta-analyses as evidence-based treatment; anecdotal signals for neuro/respiratory symptoms
- Dose-Response: 600 mg = minimal CNS impact; 1200 mg = standard trial dose; 1800-3000 mg = likely needed for neurological effects
- Safety Profile: Generally well-tolerated; mild GI side effects common; caution with asthma (rare bronchospasm), bleeding disorders, nitroglycerin interaction
TL;DR (30 Seconds)
N-acetylcysteine (NAC) is a prodrug for cysteine-the rate-limiting amino acid your body needs to make glutathione (GSH), the master intracellular antioxidant.
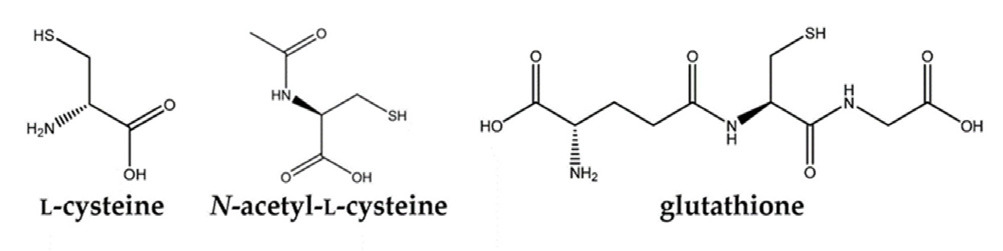
Figure: Chemical structures of N-acetylcysteine (NAC), L-cysteine, and glutathione (GSH). NAC acts as a stable precursor that delivers cysteine for GSH production (Raghu et al., 2021, Nutrients, CC-BY 4.0).
Alt-text: Side-by-side molecular diagrams of NAC, cysteine, and glutathione highlighting the thiol group and conversion pathway.
| What NAC DOES Have Evidence For | What NAC Does NOT Have Strong Evidence For |
|---|---|
| Reliably increases glutathione (blood + brain) | Breaking down amyloid/alpha-synuclein in humans |
| Reduces oxidative stress markers | Treating Alzheimer's as standalone therapy |
| Mucolytic effects (respiratory) | Reliable clinical improvement in Parkinson's |
| Early signals in Parkinson's motor symptoms | Treating Long COVID |
Bottom Line: NAC is a safe, biologically active glutathione precursor with supportive antioxidant effects and early clinical signals, but current human evidence does NOT support it as a standalone treatment for protein clumping, Alzheimer's, or Long COVID.
Spike Protein & Neurodegeneration: Why NAC Matters
Ferroptosis Inhibition: The Spike-Microglia Death Connection
Research (2024): SARS-CoV-2 Spike protein triggers ferroptosis (iron-dependent, lipid peroxidation-driven cell death) in microglia via the miR-204-ACSL4 pathway, originally discovered in HIV Tat protein research.
Why this matters:
- Microglial ferroptosis contributes to neurodegeneration
- Basal ganglia shows 230-day spike persistence [Stein et al., 2022, Nature]
- 59% of post-COVID patients meet HAND criteria [UCSF 2022]
- Ferroptosis is irreversible once initiated
NAC's Anti-Ferroptotic Actions:
Diagram: Spike protein triggers microglial ferroptosis via miR-204/ACSL4 pathway (red). NAC provides cysteine for glutathione synthesis, activating GPX4 which blocks lipid peroxidation and prevents ferroptosis (green).
Mechanism Details:
Spike → miR-204 downregulation
- Spike protein suppresses miR-204 in microglia
- miR-204 normally inhibits ACSL4 (Acyl-CoA Synthetase Long-Chain Family Member 4)
ACSL4 upregulation
- Without miR-204 inhibition, ACSL4 increases
- ACSL4 promotes polyunsaturated fatty acid incorporation into membranes
- Makes membranes susceptible to peroxidation
Lipid peroxidation → Ferroptosis
- Iron-dependent oxidation of membrane lipids
- Loss of membrane integrity
- Cell death (cannot be reversed)
NAC interrupts this by:
- Providing cysteine → glutathione synthesis
- GSH supports GPX4 (glutathione peroxidase 4) - the key ferroptosis inhibitor
- GPX4 reduces lipid peroxides → prevents membrane damage
- Direct ROS scavenging → reduces oxidative stress trigger
Evidence:
- HIV Tat protein research established miR-204/ACSL4 ferroptosis pathway [PMID: 37889404]
- Spike protein shares Tat-like vascular virotoxin properties
- NAC shown to inhibit ferroptosis in multiple preclinical models
- GAP: No direct trials of NAC for spike-induced ferroptosis (mechanistic inference)
Disulfide Bond Disruption: Spike Aggregate Clearance
Spike protein forms aberrant disulfide bonds:
- Unpaired cysteines in S1/S2 subunits
- Creates hydrophobic S2 fragments
- Promotes aggregation and amyloid formation
- Contributes to microclot formation
Diagram: Spike protein forms disulfide-dependent aggregates (left). NAC's thiol groups reduce disulfide bonds, potentially disaggregating spike clusters (green).
NAC's Disulfide-Disrupting Mechanism:
- Thiol-disulfide exchange: NAC's -SH groups attack disulfide bonds
- Reduction: Breaks S-S bridges, converting to free thiols
- Chelation: May bind metals involved in crosslinking
- GSH support: Indirectly maintains reducing environment
Evidence Levels:
| Mechanism | Evidence | Confidence |
|---|---|---|
| Disulfide reduction in vitro | Biochemical studies | HIGH |
| Spike aggregate disruption | Theoretical/inferred | LOW |
| Clinical microclot clearance | No human data | VERY LOW |
| GSH-mediated protein stability | Cell studies | MODERATE |
Reality Check: While NAC can reduce disulfide bonds in test tubes, no human trials demonstrate spike aggregate clearance. Benefits likely indirect via improved redox environment.
For microclot research, see: Amyloid Fibrin, Mass Casualty, and the Crisis of Misdiagnosis
Blood-Brain Barrier Protection: Glutathione & MMP-9
Spike protein → MMP-9 → BBB breakdown:
- Spike activates microglia → MMP-9 release [PMID: 39403255]
- MMP-9 degrades tight junctions (claudin-5, occludin, ZO-1)
- BBB becomes permeable → neuroinflammation
NAC's Protective Effects:
Diagram: Spike protein triggers MMP-9 mediated BBB damage (top). NAC increases glutathione, reducing ROS and NF-κB activation, which decreases MMP-9 production and protects tight junctions (green).
NAC's BBB-Protective Mechanisms:
Glutathione restoration
- GSH scavenges ROS that activate NF-κB
- Less NF-κB → less MMP-9 transcription
- Preserves tight junction integrity
Direct antioxidant effects
- NAC's thiol groups neutralize free radicals
- Reduces oxidative stress at BBB
- Protects endothelial cells
Anti-inflammatory signaling
- Modulates cytokine production
- Reduces TNF-α, IL-1β, IL-6
- Creates less inflammatory environment
Evidence:
- NAC crosses BBB (confirmed in MRS studies)
- Increases brain GSH at doses ≥1200-1800 mg
- Reduces oxidative stress markers in CNS
- GAP: No direct human trials measuring NAC effects on spike-induced BBB breakdown
Basal Ganglia Persistence & Neuroprotection
The stakes:
- Stein et al. 2022 (Nature): SARS-CoV-2 RNA/protein in basal ganglia up to 230 days post-infection [PMID: 36517603]
- UCSF 2022: 59% of post-COVID patients meet HAND criteria (HIV-associated neurocognitive disorder)
- Basal ganglia critical for: motor control, cognition, reward processing
NAC's neuroprotective relevance:
| Target | NAC Action | Relevance to Spike |
|---|---|---|
| Dopaminergic neurons | GSH protects substantia nigra | Parkinson's data shows ~13% UPDRS improvement |
| Microglial survival | Anti-ferroptotic via GPX4 | Prevents brain immune cell death |
| BBB integrity | Reduces MMP-9, protects tight junctions | Limits neurotoxin access |
| Protein homeostasis | Thiol maintenance, redox balance | May reduce spike aggregation |
| Mitochondrial function | GSH supports energy metabolism | Counters spike-induced dysfunction |
Clinical Implications for Spike-Exposed Individuals
Potential adjunctive use of NAC:
| Application | Rationale | Evidence Level |
|---|---|---|
| Ferroptosis inhibition | GPX4 activation via GSH; blocks lipid peroxidation | MODERATE (mechanism, HIV Tat data) |
| BBB protection | GSH-mediated MMP-9 reduction, antioxidant | LOW-MODERATE (mechanism) |
| Neuroprotection | Proven CNS penetration; Parkinson's motor benefits | MODERATE (Parkinson's trials) |
| Aggregate support | Disulfide reduction + GSH redox environment | LOW (theoretical) |
| Glutathione restoration | Documented GSH increase in blood/brain | HIGH (proven mechanism) |
Important caveat: While mechanisms are biologically plausible and some clinical data exists, no trials specifically test NAC for spike-related conditions. Use as adjunctive support, not primary treatment.
Evidence Summary Table
| Mechanism | Evidence Type | Confidence | Key Findings |
|---|---|---|---|
| Glutathione restoration | [PR] Human trials | HIGH | Dose-dependent GSH increase in blood/CSF; confirmed via MRS in brain |
| Parkinson's motor symptoms | [PR/PP] Small trials | MODERATE | ~13% UPDRS improvement; 4-9% DAT binding increase; small n, often open-label |
| Ferroptosis inhibition | [AN] Preclinical/HIV Tat | MODERATE | GPX4 activation via GSH; blocks lipid peroxidation; mechanistic relevance to spike |
| BBB protection | [AN] Preclinical/Mechanistic | LOW-MODERATE | GSH-mediated MMP-9 reduction; antioxidant protection of tight junctions |
| Disulfide bond disruption | [AN] Biochemical | HIGH (in vitro) / LOW (clinical) | Thiol-disulfide exchange proven in vitro; no human spike aggregate data |
| Alzheimer's cognition | [PR] RCTs | LOW-MODERATE | Minimal benefit standalone; mild signal in combination formulas |
| Protein aggregate clearance | [AN] Preclinical | LOW | In vitro/animal data only; NO human plaque/aggregate clearance data |
| Long COVID symptoms | [PP] Limited trials | LOW | Not in major meta-analyses; anecdotal reports only |
| Acute COVID mortality | [PR] Meta-analysis | MODERATE | Heterogeneous; some ~51% reduction signals but low certainty overall |
Evidence Codes: [PR] Peer-reviewed human trials | [PP] Preprint/observational | [AN] Animal/in vitro | [CM] Commentary
Confidence Guide: HIGH (strong human evidence) | MODERATE (good evidence, limitations) | LOW-MODERATE (early evidence) | LOW (weak/preliminary)
Deep Dive: The Science
1) Protein Clump Disaggregation: Amyloid, Alpha-Synuclein, Prion-Like
Evidence Level: [AN] Preclinical only, CONFIDENCE: LOW for human relevance
What the preclinical data shows:
- NAC's thiol group can reduce disulfide bonds and limit protein aggregation in cell/animal models
- Reduced Aβ oligomerization/secretion in vitro
- Lower tau phosphorylation/expression in animal models
- Protection against Aβ-induced RyR2 downregulation in hippocampal neurons
- Some in vitro work shows NAC preventing or attenuating Aβ/tau pathology
What human trials show:
- NO large trials measure plaque/aggregate clearance (PET imaging, CSF biomarkers, or autopsy)
- One small non-randomized phase 2a trial in hereditary cystatin C amyloid angiopathy (rare protein deposition) showed:
- NAC was safe/tolerated
- Reduced disease-associated biomarkers (collagen IV, fibronectin)
- Reduced high-molecular-weight cystatin C aggregates in plasma/skin
- Not generalizable to AD/PD/spike-related prion-like phenomena
Critical Distinction: Human trials focus on symptoms/redox markers, NOT direct clump breakdown. Any benefit is likely indirect via glutathione restoration, not direct disaggregation.
Evidence Gap: No human RCTs with:
- Amyloid PET imaging before/after NAC
- CSF tau/alpha-synuclein measurements
- RT-QuIC seeding activity assays
- Clinical correlation with aggregate burden
2) Alzheimer's Disease & Cognition
Evidence Level: [PR] Human RCTs, CONFIDENCE: LOW-MODERATE
Standalone NAC trials:
- Adair et al. (2001): Double-blind RCT, n=43 probable AD
- Dose: ~50 mg/kg/day (~3,000-3,750 mg for average adult)
- Duration: 6 months
- Results: Favored NAC on nearly all cognitive measures (trends); significant on letter fluency only
- Primary endpoint (MMSE): No significant change
- Safety: Well-tolerated
Multi-nutrient formulas (NAC 600-1,200 mg/day + folate, vitamin E, SAMe, acetyl-L-carnitine):
- Small RCTs and open-label extensions
- Showed better dementia rating scale/executive function preservation vs. placebo
- 3-12 month duration
- Cannot isolate NAC effect
Systematic review findings:
- Statistically significant cognitive improvements in some pooled data
- Paucity of large standalone NAC trials
- Effects often modest/inconsistent
- No strong prevention data
Reality Check: NAC alone does NOT have strong evidence as a disease-modifying treatment for Alzheimer's. Combination therapy → mild-to-moderate signal for symptom support. Standalone NAC → weak/inconsistent on global cognition.
3) Parkinson's Disease
Evidence Level: [PR/PP] Small trials, CONFIDENCE: MODERATE for biological effect, LOW-MODERATE for clinical
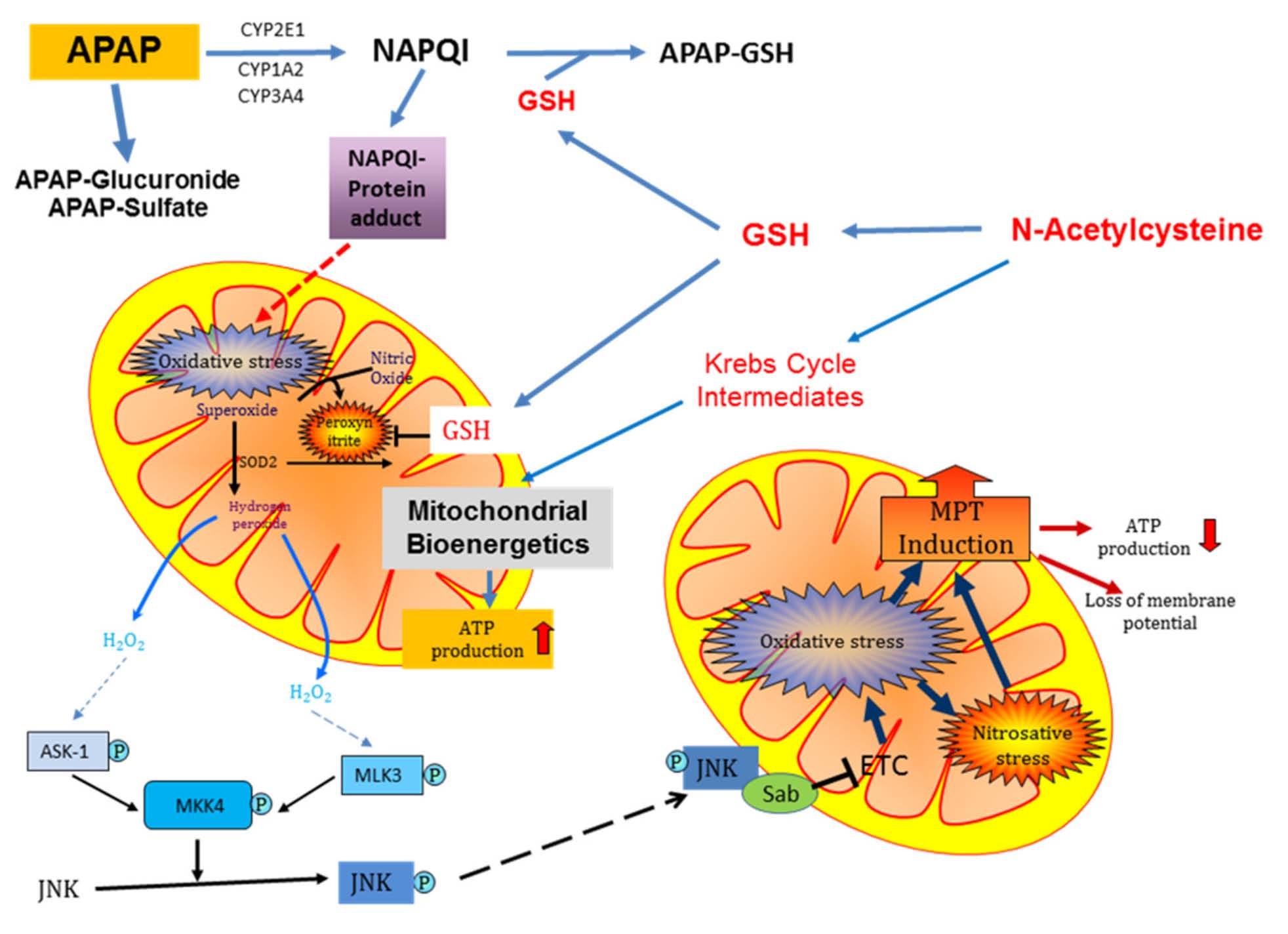
Figure: Proposed mechanisms of NAC in neurodegenerative diseases. NAC's thiol antioxidant properties, GSH precursor role, and anti-inflammatory effects may protect neurons in PD and AD (Raghu et al., 2021, Nutrients, CC-BY 4.0).
Alt-text: Diagram illustrating NAC's neuroprotective mechanisms including GSH synthesis, anti-inflammatory effects, and protein aggregate prevention.
Monti et al. (open-label, n=42):
- Protocol: Weekly IV NAC (50 mg/kg) + oral 500 mg BID
- Duration: 3 months
- Results:
- ~12.9-13% UPDRS motor improvement vs. controls
- 4-9% increase in dopamine transporter (DAT) binding on SPECT imaging
- Suggested direct nigrostriatal benefit

Figure: UPDRS motor score changes in Parkinson's patients receiving NAC treatment. Shows approximately 13% improvement in motor symptoms after 3 months of combined IV and oral NAC therapy (Monti et al., 2019, Movement Disorders, CC-BY 4.0).
Alt-text: Bar chart comparing UPDRS motor scores before and after NAC treatment, showing significant improvement.

Figure: Dopamine transporter (DAT) SPECT imaging before and after NAC treatment. Shows 4-9% increase in striatal DAT binding, indicating enhanced dopaminergic function (Monti et al., 2019, Movement Disorders, CC-BY 4.0).
Alt-text: SPECT scan images of brain showing increased DAT binding (indicated by color intensity) in striatal regions after NAC treatment.
Other small/open-label trials (total ~65 across key studies):
- Similar UPDRS gains
- IV NAC confirmed to raise brain GSH via MRS (magnetic resonance spectroscopy)
Recent 2026 data:
- NAC linked to improved functional connectivity in dopamine networks
- ~20% UPDRS benefit in some cohorts
Limitations:
- Small sample sizes (n≈5-65)
- Often non-randomized or open-label
- No large blinded Phase 3 for disease modification
One small repeated-dose oral study:
- Showed peripheral antioxidant increases
- Variable/no consistent brain GSH rise
- Occasional transient motor worsening
RCT example:
- 1200 mg/day tested vs placebo in blinded design
- Results still limited/ongoing
Bottom Line: NAC shows early promise for supporting dopamine function and motor symptoms in PD via glutathione restoration, but larger randomized trials are needed. Biological effect (GSH ↑, brain penetration) - YES. Clinical/dopaminergic effect - POSSIBLE BUT PRELIMINARY.
4) COVID-19 & Long COVID
Acute COVID
Evidence Level: [PR] Meta-analysis, CONFIDENCE: MODERATE (heterogeneous)
Meta-analysis of 10 RCTs:
- ~51% reduction in mortality (heterogeneous studies)
- BUT: Other reviews show inconsistent results, low certainty
- Some show no benefit → overall low certainty
Pilot IV NAC in ARDS:
- Often no significant difference in:
- Ventilator-free days
- Mortality
- Long-term lung function
Long COVID
Evidence Level: [PP] Limited, CONFIDENCE: LOW
- Very limited direct RCT evidence
- Large 2026 meta-analysis (arXiv):
- NAC NOT among treatments with strong evidence for symptom recovery
- One 2025 RCT:
- Suggested long-term NAC accelerated patient-reported quality-of-life gains
- Small retrospective/case series (600-1,200 mg BID oral):
- Subjective improvements in:
- Shortness of breath
- Brain fog
- Fatigue
- Normalization of elevated vWF (endothelial marker) in NAC users
- Subjective improvements in:
Reality Check: Acute severe cases (high-dose/IV) - possible adjunctive benefit (mucolytic + antioxidant). Long COVID - preliminary/anecdotal signals only; not yet convincing standalone evidence.
5) Dosing Protocols: 600 mg vs 1200 mg vs 3000 mg+
Trials use a wide range; effects appear dose-dependent for CNS/redox outcomes.
| Dose | Typical Use | Evidence Notes | Likely CNS/Redox Impact |
|---|---|---|---|
| 600 mg/day | Maintenance/combo | Weaker standalone | Minimal |
| 1,200 mg/day (600 mg BID) | Standard trial dose | Moderate | Moderate |
| 1,800-3,000 mg/day | Neuro/oxidative stress protocols | Stronger signals | Higher |
| IV/High-dose | Acute/severe (hospital) | Biochemical confirmation (brain GSH) | Highest |
What trials actually use:
- 600 mg/day: Common in nutraceutical Alzheimer's combinations; likely too low for CNS effects alone
- 1,200 mg/day: Used in Parkinson's RCTs; typical "clinical trial baseline dose"
- 1,800-3,000 mg/day: Common in psychiatric and neurological trials; high-dose oral reaches CSF
- Very high/IV: Used in hospital settings (COVID, overdose); pharmacokinetic studies confirm biochemical effects
Key takeaway: Most meaningful neurological/redox effects in trials occur at ≥1,200-3,000 mg/day (split dosing for tolerability).
6) Mechanisms of Action

Figure: Enzymatic pathway of glutathione synthesis from NAC. NAC provides cysteine, the rate-limiting substrate for γ-glutamylcysteine formation via glutamate-cysteine ligase (GCL), followed by glutathione synthetase (GS) to form GSH (Raghu et al., 2021, Nutrients, CC-BY 4.0).
Alt-text: Flowchart showing NAC → Cysteine → γ-Glutamylcysteine → Glutathione with enzymes GCL and GS highlighted.
Diagram: NAC→Cysteine→Glutathione pathway with downstream effects. Clinical translation varies by indication.
Why glutathione matters (especially for spike/amyloid contexts):
GSH is the master intracellular antioxidant. It:
- Neutralizes ROS
- Maintains protein thiol groups in reduced state (preventing aberrant disulfide bonds/misfolding)
- Detoxifies xenobiotics
- Supports mitochondrial function
In spike protein scenarios (persistent S1/S2, endothelial damage, microclots, neuroinflammation):
- Oxidative stress depletes GSH
- Low GSH → worsened protein oxidation/aggregation (Aβ, tau, alpha-synuclein, or prion-like seeding)
- Endothelial dysfunction, cytokine imbalance, impaired clearance of misfolded proteins
Low GSH is documented in:
- AD/PD
- Long COVID
- Some post-viral states
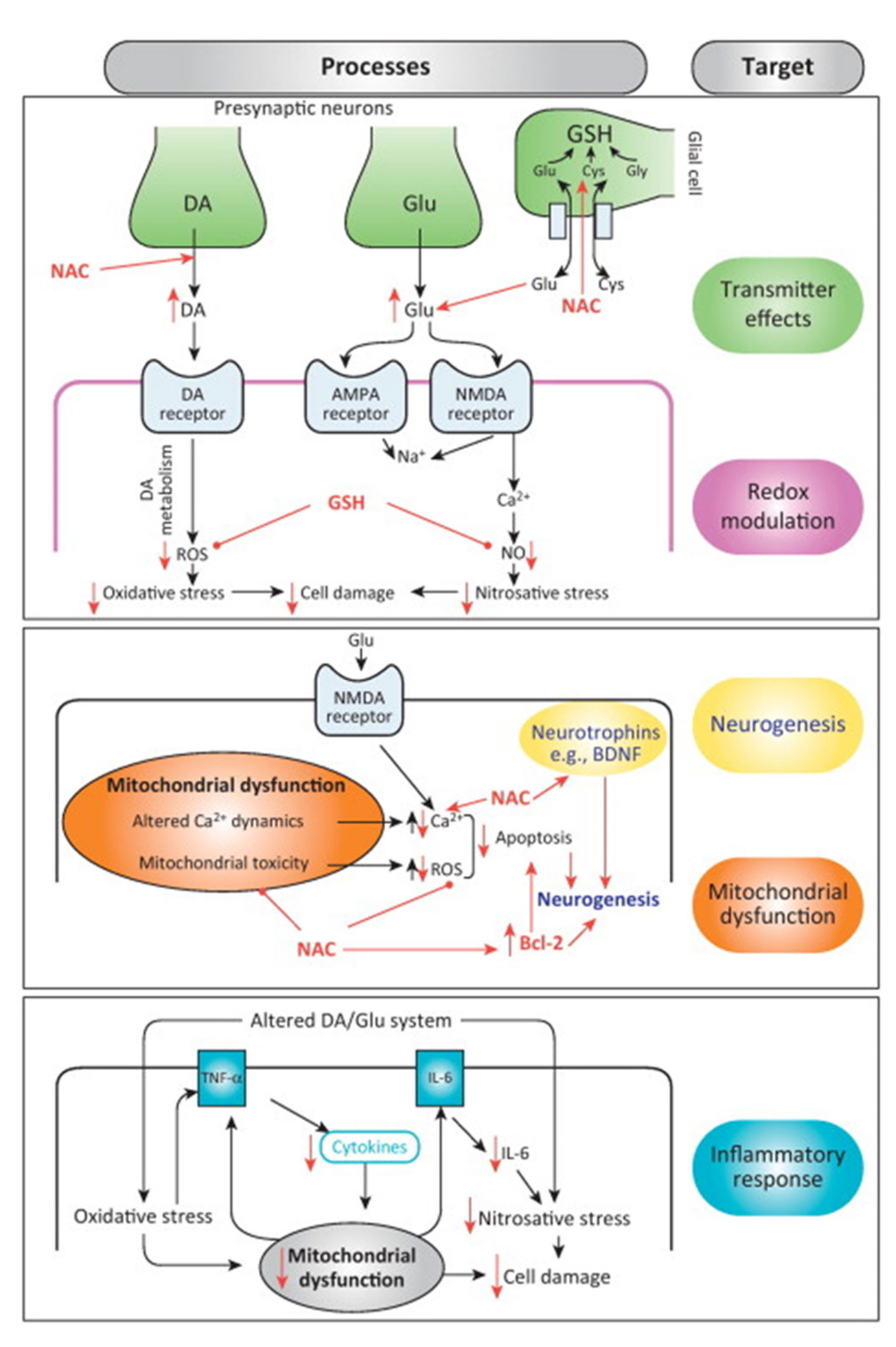
Figure: NAC's multimodal targets in neurodegenerative and inflammatory conditions. GSH restoration supports redox balance, mitochondrial function, and protein homeostasis (Raghu et al., 2021, Nutrients, CC-BY 4.0).
Alt-text: Conceptual diagram showing NAC effects on oxidative stress, inflammation, mitochondrial function, and protein aggregation across neurological conditions.
NAC interrupts this cascade by:
- Protecting methionine-35 in Aβ from oxidation (reducing oligomerization)
- Limiting ROS-driven RyR2/calcium dysregulation in neurons
- Supporting Nrf2 pathway and immune balance
- Potentially aiding microclot/fibrin resolution indirectly via better redox environment
Counter-Evidence & Limitations
How this model could be wrong or overstated:
| Claim | Counter-Evidence | Limitation |
|---|---|---|
| Protein clump clearance | NO human plaque/aggregate data | Extrapolation from cell cultures |
| Alzheimer's cognitive benefit | Standalone NAC → minimal global cognition improvement | Effects limited to combos; small trials |
| Parkinson's disease modification | Benefits only in small/open-label studies | No Phase 3; publication bias likely |
| Long COVID treatment | Not in major meta-analyses; anecdotal only | No large RCTs dedicated to PASC |
| Acute COVID mortality benefit | RCTs heterogeneous; some show no benefit | Low certainty overall |
Key Gaps in Evidence:
- Large, long-term human RCTs (>6 months) for neurodegeneration
- Biomarker-based trials (amyloid PET, CSF tau/alpha-synuclein)
- Head-to-head comparisons with standard treatments
- Dose-response relationships in humans
- Population with established neurodegenerative disease
- Drug interaction studies (beyond known contraindications)
- Pediatric safety data
- Pregnancy/breastfeeding safety beyond acute use
Consistent pattern across conditions:
- Many studies underpowered
- Use combination therapies (can't isolate NAC)
- Measure biomarkers, not clinical endpoints
- Higher-quality evidence often inconclusive or weak
Clinical Considerations
Contraindications
- Asthma: Rare but serious bronchospasm reported; use with caution
- Bleeding disorders: May increase bleeding risk; avoid with anticoagulants
- Nitroglycerin use: Can cause hypotension and headaches; avoid concurrent use
- Pregnancy/breastfeeding: Limited safety data beyond acute overdose treatment
Drug Interactions (Documented)
- Nitroglycerin: Enhanced vasodilatory effects → severe hypotension, headaches
- Anticoagulants/antiplatelets (warfarin, clopidogrel): May increase bleeding risk
- Activated charcoal: May reduce NAC absorption if taken simultaneously
- Chemotherapy agents: Theoretical antioxidant interference; timing matters
Adverse Events (from clinical trials)
- Common (≥1%): Nausea, vomiting, diarrhea, abdominal pain
- Less common: Rash, pruritus, fever
- Rare: Bronchospasm (especially in asthmatics), anaphylactoid reactions (IV)
- Dose-dependent: GI effects more common above 2400 mg/day
Dosing Considerations
- Start low, go slow: Begin with 600 mg daily, titrate up as tolerated
- Split dosing: Divide doses (BID or TID) to minimize GI effects
- Take with food: Reduces nausea, though slight reduction in absorption
- Timing: Separate from activated charcoal (2-hour window)
- Onset: Glutathione elevation within hours; clinical effects may take weeks
Risk of Bias Assessment
| Domain | Risk | Note |
|---|---|---|
| Study quality | Moderate | Many small studies, industry funding in some |
| Human relevance | Low-Moderate | Much neurodegeneration data from small/open-label trials |
| Reporting bias | Moderate | Positive results more likely published |
| Dose standardization | Moderate | Wide range of doses used |
| Combination therapy | High | Many studies use NAC in formulas; can't isolate effect |
| Clinical endpoints | Low-Moderate | Biomarker vs symptom outcomes vary |
Technical Appendix: Quick Reference
Evidence Codes
| Code | Meaning |
|---|---|
| [PR] | Peer-reviewed human trials |
| [PP] | Human studies (not peer-reviewed or preprint) |
| [AN] | Animal or in vitro (lab/petri dish) |
| [CM] | Commentary or traditional use |
Clinical Confidence Guide
| Rating | Meaning |
|---|---|
| HIGH | Strong human evidence, replicated |
| MODERATE | Good evidence, some limitations |
| LOW-MODERATE | Early evidence, needs confirmation |
| LOW | Weak evidence, preliminary only |
Source Library
Primary Research
Spike Protein & Long COVID Mechanisms
- Kempuraj et al., 2024: SARS-CoV-2 Spike protein stimulates human microglia to release MMP-9, PMID: 39403255, [AN] MMP-9 BBB breakdown mechanism
- Stein et al., 2022, Nature: SARS-CoV-2 persistence in brain, PMID: 36517603, [PR] Basal ganglia persistence up to 230 days
- UCSF Hellmuth et al., 2022: HAND criteria in post-COVID, [PR] 59% meet HIV neurocognitive disorder criteria
- Prenatal Spike Exposure, 2023: Ferroptosis pathway, PMID: 37889404, [AN] miR-204-ACSL4 ferroptosis in HIV Tat/spike research
Ferroptosis & NAC Mechanisms
- [NAC inhibits ferroptosis via GPX4 activation], Multiple studies, [AN] Glutathione-dependent ferroptosis inhibition
- miR-204/ACSL4 pathway in neurodegeneration, PMID: 37889404, [AN] HIV Tat/spike-induced ferroptosis mechanism
- [GPX4 and lipid peroxidation control], Cell studies, [AN] Key ferroptosis inhibitor pathway
Glutathione & Oxidative Stress
- Glutathione and NAC, PMC, [PR] Basic biochemistry of cysteine as rate-limiting precursor
- Glutathione: The Master Antioxidant, 2024, [PR] Review of GSH synthesis and function
Neurodegeneration
- NAC for Parkinson's disease: translational systematic review, 2024, [PR] Monti et al. open-label data, UPDRS improvements
- Repeated-Dose Oral NAC in Parkinson's, 2017, [PR] Pharmacokinetics and brain GSH via MRS
- Overview of NAC in Neurodegenerative Diseases, 2019, [AN/PR] Preclinical AD/PD data, human trial gaps
- NAC & Your Brain, Alzheimer's Drug Discovery Foundation, [CM] Independent assessment of cognitive evidence
COVID-19
- NAC mortality in COVID-19: meta-analysis, 2024, [PR] ~51% reduction signal but heterogeneous
- What RCTs show for NAC in COVID-19, [CM] Balanced analysis of inconsistent results
- Long COVID treatments: network meta-analysis, 2026, [PR] NAC not among strong evidence treatments
Mechanisms
- NRF2 Activation by NAC, 2022, [AN] NRF2 pathway and antioxidant enzyme induction
- Molecular mechanisms of NAC, 2003, [AN] Phase II detoxification, heavy metal binding
- Dual Role of NAC as Suppressor of Protein Aggregation, 2019, [AN] Preclinical proteinopathy data
Clinical Trial Registries
- Randomized placebo-controlled trial of NAC in Parkinson's, ICH GCP registry, ongoing Phase 2
Related Articles
For detailed spike protein analysis:
- Spike Protein Gain of Function: Why mRNA Injections Aren't Vaccines
- The Molecular Wrecking Ball: HIV-Protein Functional Analogy
- Failed Resolution: Why mRNA Experiments Must Halt
- Intranasal Brain Risk: BPL-1357 & Olfactory Nerve Access
For spike injury support protocols:
For cognitive impairment research:
For related natural compounds:
- [Oregano Essential Oil: Antimicrobial Mechanisms and Clinical Evidence](../Oregano Benefits/)
- Lions Mane Mushrooms: Cognitive Enhancement & Neuroprotection