Table of Contents
TL;DR
The mRNA vaccine platform won a Nobel Prize for brilliant engineering—specifically, making synthetic mRNA stable enough to work as a drug. But between clinical trials and mass production, something fundamental changed.
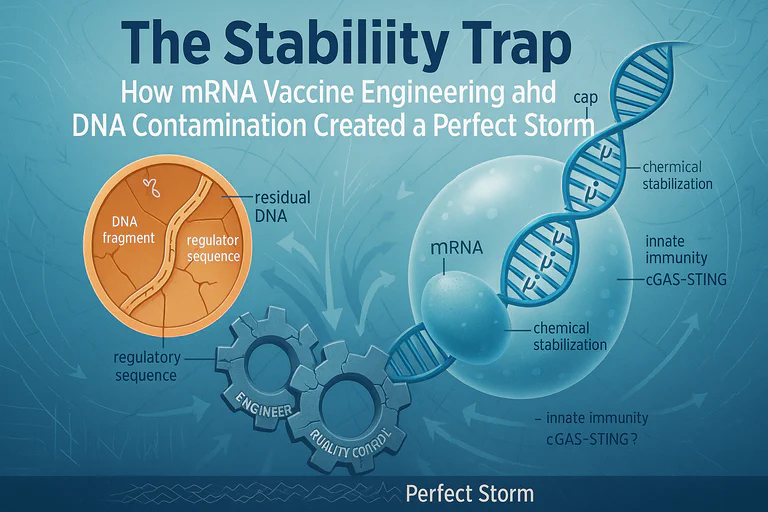
The problem in plain terms: The vaccine billions received was not the same one tested in trials. It contains bacterial DNA with active viral promoters—contamination that internal regulatory documents show alarmed scientists while public assurances dismissed it.
Why this matters: This DNA isn't inert. The m⁶A methylation pattern confirms it came from E. coli. The SV40 promoter is unmethylated and biologically active. Together, they can trigger chronic immune activation, may interfere with antibody production through somatic hypermutation disruption, and carry integration risks that weren't adequately studied.
The evidence: 9+ independent labs across four continents have confirmed contamination levels up to 627-fold above regulatory limits. The batch variance (815-fold) reveals systemic manufacturing control failure.
What we don't know: The long-term clinical consequences of chronic DNA exposure via lipid nanoparticles. Population-level data won't be visible for 5-10 years if solid tumors are the endpoint. But the biological mechanisms for harm are documented, and the regulatory transparency gap is undeniable.
Note: This is educational content, not medical advice. Clinical decisions belong with qualified healthcare professionals.
The Engineering That Won a Nobel Prize—and Its Unintended Price
Drew Weissman and Katalin Karikó solved a fundamental problem: making mRNA stable enough to work as a drug. The m1Ψ (pseudouridine) modification was brilliant—it made synthetic mRNA "invisible" to our innate immune system. Lipid nanoparticles provided the armored delivery system.
But in medicine, every solution begets new questions. The very modifications that stopped our bodies from breaking down synthetic mRNA may have created something we didn't anticipate: a vaccine that doesn't clear on schedule, leading to prolonged spike production and cascading biological consequences.
The Switch Nobody Announced: Process 1 vs Process 2
Here's what happened: Pfizer's clinical trials used "Process 1" manufacturing. The version billions received came from "Process 2." Were they the same product?
We don't know—because the planned safety comparison vanished.
Pfizer's original trial design included a cohort receiving both Process 1 and Process 2 material for direct comparison. Then in September 2022, Protocol Amendment 20 removed this objective just as Process 2 dominated global supply. When researchers filed Freedom of Information requests asking how equivalence was demonstrated, regulators acknowledged the question but provided no analytical data.
For detailed analysis of manufacturing changes, see Pfizer Process 1 vs Process 2: What Changed, What's Measured, and What Remains Uncertain
The Contamination: It's Not Just DNA, It's Biologically Active DNA
Multiple independent labs across four continents have found significant levels of plasmid DNA fragments in vaccine vials. But forensic sequencing reveals this isn't just about quantity—it's about biological activity.
The Global Laboratory Consensus (9+ teams confirming):
- McKernan (USA) – Found residual plasmid DNA at 10-843 ng/dose
- Buckhaults (USA) – Cancer geneticist confirmed contamination and oncogenic risk in Senate testimony
- König/Kirchner (Germany) – Reported 3,600-5,340 ng/dose (500x over limit)
- Kämmerer (Germany) – Identified SV40 promoter/enhancer and LNP-mediated delivery
- Raoult (France) – Found full plasmid recovery in vials
- Plus additional teams in the USA and Europe (some choosing anonymity)
The Peer-Reviewed Quantification
Speicher et al. (2025) provided the first comprehensive analysis of 27 mRNA vaccine lots:
- Residual DNA exceeded EMA/FDA limits by up to 627-fold
- SV40 promoter-enhancer detected at 0.25–23.72 ng/dose in Pfizer vials
- Oxford Nanopore sequencing revealed fragments up to 3.4 kb, large enough for functional integration
- 815-fold variance in dsDNA levels between batches
- Mean fragment length ~214 bp with maximum fragments up to 3.5 kb
- Direct correlation between higher DNA levels and increased VAERS adverse event reports

DOI: 10.1080/08916934.2025.2551517, PMID: 40913499
The Forensic Smoking Gun
Sequencing of Pfizer lot FL8095 reveals the contamination is more dangerous than initially understood:
- Bacterial Methylation Signature: N⁶-methyladenine (m⁶A) methylation in GATC motifs—the unmistakable fingerprint of E. coli "Dam" methylase
- Incomplete Linearization: Evidence of plasmid fragments that failed to be properly linearized, with potential for circular plasmid forms to persist
- "Naked" SV40 Enhancer: The SV40 promoter/enhancer region is notably unmethylated, meaning it's fully active



We're not dealing with generic "DNA fragments." We're dealing with specifically modified, immunostimulatory, bacterial plasmid DNA with active viral promoters, delivered directly into human cells via LNPs.
Forensic sequencing data, Source PDF, m6A methylation analysis (Kevin McKernan)
For comprehensive laboratory findings, see SV40 DNA Signals in COVID-19 mRNA Vaccine Vials
The Regulatory Crisis: Private Alarm, Public Reassurance
Internal Health Canada emails obtained through investigative work reveal a regulatory crisis concealed from public view:
Pfizer's admission: "Yes, because Pfizer did not identify the presence of SV40 promoter enhancer on the plasmid template used to produce mRNA"
Regulatory alarm bells:
- "SV40 must be avoided!", Clear safety principle violation
- "Remedy the situation before Fall 2024 vax campaign", Urgent timeline
- "They do not seem to care much at this moment", Frustration with Pfizer's response
- "Fragment size is related to the probability of integration", Direct genotoxicity discussion
- "References: None" beside "minimal safety risk" claim, Unsubstantiated assurance
The Manufacturer Knew: Moderna's Patent Warnings
Moderna's own patents from years before COVID-19 explicitly warned:
- "DNA fragments resulting from the mRNA production process need to be removed"
- "Because the remaining DNA fragments may cause a patient to develop cancer"
- "qPCR can't measure small DNA fragments, yet we have to use another method to quantify them"
- "Some of the introduced DNA may be incorporated into the genome of the cell and inherited by the offspring"
The risks were well-understood, documented, and explicitly warned against—years before the pandemic vaccine rollout.
How Harm Could Happen: Multiple Plausible Mechanisms
The combination of persistence and biologically active contamination creates several pathways to harm.
Mechanism 1: Chronic Innate Immune Activation
The m⁶A-methylated bacterial DNA is a potent ligand for innate immune sensors (cGAS-STING and TLR9). These sensors trigger pro-inflammatory cascades (Type I Interferons, NF-κB, IL-6, TNF-α). Because the stabilized mRNA/LNP platform persists, this alarm signal isn't transient—it's chronic.
Consequences: Oxidative stress, mitochondrial damage, impaired DNA repair, immune exhaustion, loss of self-tolerance.
Mechanism 2: Somatic Hypermutation Disruption
A January 2024 preprint reveals an even more concerning mechanism: the SV40 enhancer functions as a somatic hypermutation targeting element, hijacking AID (Activation-Induced Cytidine Deaminase) enzyme. This can redirect antibody diversation, potentially causing impaired immune responses or oncogenic mutations.
DOI: 10.1101/2024.01.09.574829
What this means: If SV40 hijacks the AID enzyme, your body's ability to produce diverse, effective antibodies may be compromised. Immunologist Jessica Rose notes: "If we have impaired antibody responses, we have a broken immune system."
Mechanism 3: Nuclear Localization and Integration Risk
The SV40 promoter-enhancer functions as a nuclear localization signal (NLS)—a molecular "passport" that actively transports DNA into the cell nucleus. Once inside, DNA fragments can integrate into chromosomes via non-homologous end joining, retrotransposon-mediated insertion, or double-strand break repair mechanisms.
Precedent: The FDA's Keith Peden established the 10 ng/dose DNA limit specifically to minimize insertional mutagenesis risk. The 627-fold exceedance in some lots represents a catastrophic regulatory failure.
SV40 classification: The International Agency for Research on Cancer (IARC) classifies SV40 as a Group 2A carcinogen (probably carcinogenic to humans) based on tumor induction in animal models, detection in human tumors, and in vitro transformation of human cells.
The Persistence Evidence: Spike Stays Longer Than Expected
Foundational Studies
Röltgen et al. (2022) - Yale Study: Showed mRNA and spike antigen persist in germinal centers of lymph nodes for at least 60 days post-vaccination.
Patterson et al. (2021): Found S1 sub-units of the spike protein in monocytes for up to 15 months.
Cell: DOI: 10.1016/j.cell.2022.01.018, Frontiers: DOI: 10.3389/fimmu.2021.746021
Epidemiological Signals
Ueda et al. (2024) analyzed age-adjusted cancer mortality in Japan following widespread mRNA vaccination, finding 7,162 excess cancer deaths in 2022 (95% CI: 4,786–9,522) with increases observed after the third mRNA-LNP dose.
Important caveats:
- Temporal association does not prove causation
- Multiple confounders exist (delayed screenings, pandemic stress, healthcare disruption)
- The article has been retracted by the publisher—findings are no longer considered reliable
Bottom line: Any cancer mortality signal requires independent confirmation and 5-10 year lag time for solid tumor detection. U.S. cancer registry data (2021-2022) showed no significant increase in overall incidence, but early signals may not yet be visible.
The Perfect Storm: How Everything Converges
When you combine all these factors, a troubling picture emerges:
Engineering Persistence Platform:
- Stabilized mRNA (m1Ψ modification)
- Efficient LNP delivery
- Prolonged spike production
Biologically Active Contamination:
- Bacterial methylated DNA (m⁶A signatures)
- Active SV40 viral promoters
- Plasmid DNA with integration potential
Immune System Interference:
- Chronic innate immune activation (cGAS-STING/TLR9)
- Somatic hypermutation disruption (AID targeting)
- Antibody diversity compromise
Systemic Regulatory Failure:
- Manufacturing process changes without validation
- Internal regulatory alarm without public disclosure
- Removal of planned safety comparisons
- Manufacturer prior knowledge without adequate mitigation
The Regulatory Failure: Standards Ignored
EMA ICH Q5B – Standards for biologics typically allow <10 ng/dose of residual DNA and explicitly forbid sequences with known oncogenic activity.
The Transparency Gap:
- No public release of Process 1 vs Process 2 comparability data
- Internal regulatory concerns concealed from public view
- Independent lab findings dismissed without GMP-grade replication
- Manufacturer patents showing prior knowledge ignored in risk assessment
Regulatory Divergence:
The SV40 contamination created an unprecedented regulatory split:
- Florida Department of Health (January 2024): Halted mRNA vaccine use, cited SV40 as potentially oncogenic, demanded biodistribution and integration studies
- FDA (December 2023): Claimed SV40 fragment is "inactive" without replication origin, asserted no oncogenic risk
- TGA Australia (December 2024): Acknowledged SV40 presence, denied cancer link based on lack of epidemiological evidence
The dispute: FDA says the promoter fragment is harmless without the full viral genome. Critics argue the promoter itself is the oncogenic risk via NLS-mediated integration, not viral replication. This isn't resolved—Florida halted use while federal agencies maintained safety.
What the Evidence Actually Shows
| Claim | Evidence | Grade |
|---|---|---|
| DNA contamination exceeds regulatory limits | Speicher et al. 2025: Up to 627-fold exceedance, 100% of lots contaminated (27 lots analyzed) | High |
| SV40 promoter present in vials | McKernan et al.: Detected across multiple independent laboratories globally | High |
| Bacterial m⁶A methylation confirms E. coli origin | McKernan et al. 2025: N⁶-methyladenine in GATC motifs, bacterial fingerprint | High |
| Spike persists months post-vaccination | Röltgen et al. 2022: mRNA and spike detected ≥60 days in lymph nodes | Moderate-High |
| SV40 enhancer hijacks somatic hypermutation | bioRxiv 2024 preprint: SV40 acts as SHM targeting element in vitro | Moderate (mechanistic) |
| Japan excess cancer mortality proves causation | Ueda et al. 2024: Article retracted by publisher; findings no longer reliable | Invalid |
Key Takeaway: DNA contamination is confirmed fact. SV40 biological activity is mechanistically plausible. Population-level clinical impact remains uncertain. What's clear is that regulatory closure of legitimate questions undermines trust—transparency has been inadequate.
What Both Sides Agree On
Even amidst heated disagreement, there's consensus on several points:
- DNA contamination exists and exceeds limits
- SV40 sequence is present
- More long-term surveillance is needed
- Transparency has been inadequate
Biomarker Tracking: For Those Concerned
If you're experiencing persistent symptoms or are concerned about vaccine exposure, tracking biomarkers over time can provide actionable data.
How to use: Repeat every 8–12 weeks while symptoms evolve. Look for directional trends, not single "perfect" numbers. Most advanced testing requires specialty labs.
| Panel | Biomarker | Why It Helps | Availability |
|---|---|---|---|
| Spike Persistence | Circulating spike protein (LC-MS/MS) | Direct detection of persistent antigen | Research setting |
| Anti-spike antibody ratios | Persistent exposure vs declining immunity | Standard labs | |
| Inflammation | hs-CRP, IL-6, TNF-α | Chronic immune activation burden | Standard labs |
| Autoimmunity | ANA, anti-phospholipid antibodies | Self-attack marker development | Standard labs |
| DNA Damage | γH2AX (phosphorylated histone) | DNA double-strand break marker | Research/specialty labs |
| 8-OHdG | Oxidative DNA damage marker | Specialty labs | |
| Cellular Health | LDH, ferritin | General cell turnover/inflammation | Standard labs |
| D-dimer, fibrinogen | Coagulation/microclot assessment | Standard labs |
Reality check: Absence of testing does not mean absence of risk. Symptoms and clinical judgment remain important. Work with a knowledgeable clinician for interpretation.
A Plain Language Summary
If technical details feel overwhelming, here's the simple version:
mRNA vaccines were a brilliant engineering solution that won a Nobel Prize. But something went wrong between clinical trials and mass production. The vaccine billions received was not the same one tested in trials. It contained bacterial DNA with active viral promoters—contamination that regulatory scientists privately called alarming while publicly dismissing it.
This DNA is not inert. It can trigger chronic immune activation and may interfere with how your body makes antibodies. The spike protein itself persists longer than expected in some people.
What this means: We're in uncharted territory. The "safe and effective" narrative does not match the documented reality. More research is needed. More transparency is required. Your health autonomy matters.
Think of it like a car: The engineering triumph is a revolutionary engine that wins awards. The problem is the production version was built differently than the prototype, with unauthorized parts that engineers privately warned about but manufacturers used anyway. The result is cars being recalled while regulators say "drive it anyway, it's probably fine" while some mechanics find dangerous problems.
If you're experiencing symptoms: Fatigue that doesn't improve with rest, brain fog, racing heart, new allergies, muscle and joint pain, "flare-ups" triggered by exertion, feeling "wired but tired"—these are real symptoms that deserve real investigation, not dismissal.
Frequently Asked Questions
Should I get boosted if I've had reactions?
That's your decision. But consider: (1) Prior reactions increase risk of future reactions; (2) No data exists on safety for those with existing complications; (3) The burden of proof should be on demonstrating safety, not assuming it.
Can I test for DNA contamination or spike persistence?
LC-MS/MS testing can detect spike protein in research settings. Most clinical labs don't offer this. Antibody ratios and inflammatory markers provide indirect clues. Specialty testing is emerging but not widely available.
What about natural immunity vs vaccination?
Natural infection provides broader mucosal immunity and longer-lasting memory cell responses than mRNA vaccination. Hybrid immunity (infection + vaccination) shows strongest responses in some studies. Individual circumstances vary.
Is the SV40 contamination the same as polio vaccine contamination?
Different context, same virus family. SV40 in polio vaccines (1955-1963) was later linked to rare cancers. The mRNA SV40 fragment is a promoter-enhancer sequence, not the whole virus. Both represent unintended biological contaminants with uncertain long-term effects.
Will this give me cancer?
We don't know. Biological mechanisms are plausible (SV40 integration, chronic inflammation). But solid tumor development typically takes 5-10 years. What we know: the risk is not zero. What we don't know: the actual magnitude. That's why long-term surveillance matters.
Related Investigations
For deeper exploration of specific aspects:
SV40 DNA Signals in COVID-19 mRNA Vaccine Vials, Comprehensive analysis of independent laboratory findings across international teams
Pfizer Process 1 vs Process 2: What Changed, Investigation into manufacturing changes and removed safety comparisons
DNA Contamination & mRNA Vaccine Biology: Curated Reference Roadmap, Primary sources, regulatory documents, and scientific studies
Conclusion: From Triumph to Trap
The emerging picture reveals a perfect storm of intersecting factors.
What we know:
- DNA contamination is real, widespread, and confirmed by global scientific consensus
- The contamination is biologically active with bacterial methylation and functional SV40 promoters
- Internal regulators were alarmed while maintaining public assurances
- Manufacturers documented these risks years before the pandemic
- Multiple biological mechanisms exist for potential harm
What we don't know:
- The full clinical impact of chronic DNA exposure via LNPs
- The population-level effects of somatic hypermutation disruption
- The long-term consequences of persistent spike protein and immune activation
- Whether early cancer mortality signals represent true causality or confounding
- The integration frequency in human tissues and which cell types are most affected
The burden of proof has decisively shifted. It's no longer sufficient to say "no evidence of harm"—we need robust evidence of safety in light of these documented contamination issues, identified biological mechanisms, and regulatory transparency failures.
The story of mRNA vaccines is evolving from one of pure triumph to a complex case study in how engineering solutions can create unintended biological consequences when combined with quality control failures and regulatory breakdowns.
Key References
Primary Research:
Speicher et al. (2025). Residual DNA and SV40 sequences in mRNA vaccine lots. Autoimmunity. DOI: 10.1080/08916934.2025.2551517, PMID: 40913499
Röltgen et al. (2022). mRNA and spike antigen persistence in lymph nodes. Cell. DOI: 10.1016/j.cell.2022.01.018
Patterson et al. (2021). S1 sub-unit persistence in monocytes. Frontiers in Immunology. DOI: 10.3389/fimmu.2021.746021
SV40 somatic hypermutation mechanism. bioRxiv preprint. DOI: 10.1101/2024.01.09.574829
Regulatory Documents:
EMA ICH Q5B guideline on residual DNA limits, EMA guideline
Health Canada FOI emails on SV40 contamination, Scoops McGoo reporting
Florida Department of Health guidance (January 2024), Archived PDF
FDA response on SV40 safety (December 2023), Agency PDF
Independent Laboratory Analyses:
McKernan et al. Forensic sequencing of Pfizer lot FL8095. Zenodo. doi:10.5281/zenodo.17281691
McKernan et al. RNA:DNA hybrids in vaccine manufacturing. Journal of Independent Medicine. https://journalofindependentmedicine.org/articles/v02n01a04/
This analysis synthesizes emerging scientific evidence from peer-reviewed studies, regulatory documents, manufacturer patents, and independent laboratory analyses. It is presented for informational and discussion purposes to advance scientific transparency and public understanding. It is not medical advice. Consult healthcare professionals for medical decisions.